Alzheimer’s Biomarkers
Biomarkers for Alzheimer’s disease are measurable indicators of changes in the brain that signal the presence or risk of the disease. Common biomarkers include beta-amyloid plaques, tau tangles, and signs of neurodegeneration or brain inflammation. These can be detected through brain scans, cerebrospinal fluid (CSF), or blood tests.
The three main types of Alzheimer’s biomarkers
What it is
Beta-amyloid plaque is a sticky buildup of protein fragments that clump together between nerve cells in the brain. These plaques are a hallmark of Alzheimer’s disease and are believed to interfere with cell-to-cell communication, trigger inflammation, and contribute to the death of brain cells over time.
How to detect
What are they
Tau tangles, also known as neurofibrillary tangles, are twisted fibers of abnormal tau protein that build up inside brain cells. In Alzheimer’s disease, these tangles disrupt the transport of nutrients and signals within neurons, leading to cell damage and cognitive decline.
How to detect
What it is
Inflammation in Alzheimer’s disease is studied using blood tests, cerebrospinal fluid (CSF) analysis, and advanced brain imaging techniques like PET scans. Researchers look for elevated inflammatory markers such as cytokines or proteins like YKL-40 and GFAP, which may indicate brain inflammation linked to neurodegeneration.
How to detect
Interactive learning guide: The Science of Alzheimer’s
Click on our interactive guide on the Science of Alzheimer’s to learn more.
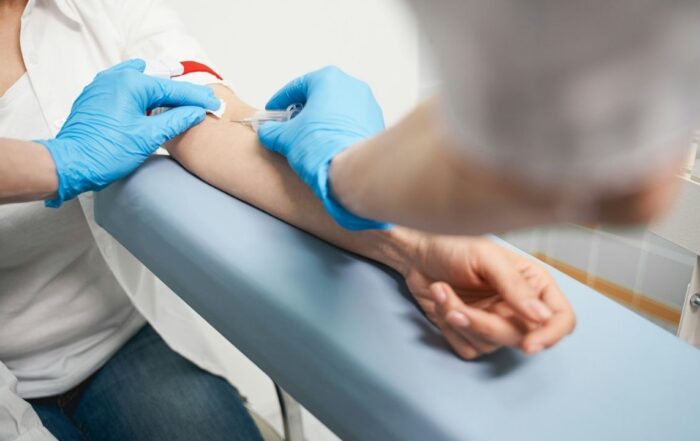
The role of blood tests in Alzheimer’s diagnosis
Blood Tests for Alzheimer’s
Blood tests for Alzheimer’s disease are an emerging diagnostic tool that aim to detect biological markers — or biomarkers — associated with the disease. These tests are not yet a stand-alone diagnostic method but are increasingly used to support diagnosis,
screen for risk, or rule out other causes of cognitive decline.
Specific biomarkers detected
Interactive learning guide: Blood-based biomarkers for Alzheimer’s disease
Click on our interactive guide below to learn more.
Explore our lifestyle learning guides on brain health
Click on a topic below to view the guide.
Share your diagnosis story
Have you experienced symptoms that left you wondering if they were part of normal aging or something more serious? Did you reach out to a doctor for help shedding light on what was going on?
Tell us your about your journey to diagnosis. We’re looking for stories about how people have sought answers about their cognitive health, managed symptoms, and more.
FAQs about Alzheimer’s biomarkers
Blood tests are not yet a stand-alone diagnostic method but are increasingly used to support diagnosis, screen for risk, or rule out other causes of cognitive decline.
Researchers don’t have enough data on what a positive biomarker test means in otherwise healthy individuals. Since these tests were designed for people with cognitive problems, there is a high likelihood of false positives. Speak with a dementia specialist about the results or consider enrolling in a study or trial that will help scientists understand what this result means.
A positive biomarker result is necessary but not sufficient. The two FDA-approved disease-modifying drugs — Leqembi and Kisunla, which is also designed to target amyloid — are approved for people in the early stages of Alzheimer’s disease who test positive for amyloid buildup. In some countries, people who carry two copies of the ApoE4 gene are excluded from eligibility because they have a higher risk of side effects, including brain swelling and bleeding.